CPN is honored that the Byers family has agreed to share and reflect on their experience of having their son Will participate in two very different clinical trials. In Part One of this narrative, Will’s mother, Valerie sets the stage for Will’s participation in the first trial and discusses the clinical trial process, in particular the complex decisions they faced and the myriad of emotions that accompanied them.
The Call
The vibration in my pocket started while I was lying on my back, the dentist massaging my gums as he inserted multiple shots of Novocaine into the back of my mouth. My phone made the familiar buzzes of an incoming call and then a short buzz to let me know I’d missed the call. Another single buzz followed, letting me know there was a voicemail.
“Can you feel this? Does this hurt?”
Oddly enough, the root canal, the repercussion of an old filling that had cracked, was the least of my worries. The truth was that, while I couldn’t feel anything in my mouth, everything about me was hurting. It was the worst week of my life. My chatty, loving, creative, and joy-filled 4-year old son, Will, had been given a death sentence when geneticists confirmed that he had Sanfilippo Syndrome. Sanfilippo Syndrome is a rare genetic condition in which a person is missing a critical enzyme used in ridding the body of cell waste. This waste then builds up in every system of the body and causes cell damage and death. It would eventually rob my son of his ability to talk, eat, and walk and then take his life. To look at him running around outside in the warmth of our Texas spring singing songs from his favorite cartoons inspired both disbelief – how is this even possible?…… and heartbreak – he will lose his ability to do this and I will lose him.
 There is no cure or treatment for Sanfilippo Syndrome. It’s not for want of trying. Since the discovery of the condition in 1963, parents and scientists have been working against time to try to find a mechanism to slow, stop, and/or reverse the disease’s effects. However, as with many rare diseases, the money and interest in combatting the condition is limited. Work comes from the grassroots efforts of affected families and dedicated researchers. While the effort and commitment level in these parties is high, the reality is that without large-scale public funding and momentum, many children live and die with no hope of anything other than the standard progression of the disease.
There is no cure or treatment for Sanfilippo Syndrome. It’s not for want of trying. Since the discovery of the condition in 1963, parents and scientists have been working against time to try to find a mechanism to slow, stop, and/or reverse the disease’s effects. However, as with many rare diseases, the money and interest in combatting the condition is limited. Work comes from the grassroots efforts of affected families and dedicated researchers. While the effort and commitment level in these parties is high, the reality is that without large-scale public funding and momentum, many children live and die with no hope of anything other than the standard progression of the disease.
With so many rare diseases lacking official treatments, the hope of many rare disease families comes in the form of clinical trials. They seek out the researchers specializing in the rare condition, talk to them, see if there is any option for clinical trial enrollment. If there is no clinical trial, then the families look to see where the research is at and begin fundraising to help support the current work with the goal of getting the research to a clinical trial point. Largely, they do this work with the hope that their child or family member may be able to participate should a trial come to fruition.
We were no different. Although newly diagnosed and grieving the dreams that we had for our son’s life, we jumped in to trying to wrap ahead around this disease and look for something, anything, we could do to help Will. One of the first things we did was begin to connect with other families. We wanted to know what they knew, to understand the journey they had been taking and that we were just starting. These families quickly became our lifeline. They understood exactly where we were and what we were feeling in a way no one else in our lives could. One family even ended up giving us a chance at life for Will.
Leaving the dentist, root canal over and my mouth as numb as my soul had been for a week, I took out my phone to listen to my voicemail. “Mrs. Byers, this is Dr. – with the University of Minnesota Masonic Children’s Hospital. I’ve heard from another family that your son, Will, is 4-years old and was recently diagnosed with Sanfilippo Syndrome, Type B. I just wanted to let you know that we are currently recruiting for a clinical trial for his condition and have limited spots remaining. Please call me back and let me know if you’d be interested in having your son screened for the trial. Thank you.”
My heart stopped and a small seed took root in that stillness and began to grow a feeling I hadn’t felt that week: hope.
The Chance
Our phone call was an oddity – you just don’t get diagnosed with Sanfilippo Syndrome and then get a phone call that same week asking you to come screen for a clinical trial. It was simply the luck of having connected to the right people at the right time. In this case, we had connected with another family screening for a different clinical trial at the same location just as a clinical trial for our son’s subtype of Sanfilippo was actually recruiting for participants. Sanfilippo Syndrome, especially in 2015, had an extremely low number of clinical trials recruiting. And by small, I mean two – 0ne for the more common subtype,Type A and one for the more rare Type B, which is the subtype Will has.
 The more likely process for rare disease families is to stalk the clinical trial website daily investigating the listed trials, trying desperately to understand unfamiliar research terminology and gain an unofficial medical degree overnight. You watch for “Active, not recruiting” to change to “Recruiting.” You call and email the clinical trial coordinators and then call and email again when they don’t respond back to you soon enough. Then you have your local doctors call and email on your behalf, hoping they will get a response. You struggle to understand, when your loved one is literally dying a little more every second of every day, how clinical trial administrators can just react without your sense of life-or-death urgency. You curse the very guidelines and bureaucracy that you know are designed to keep participants safe for delaying the one thing that could actually make a difference in your loved one’s prognosis. You see the limited number of spots available and feel guilty about competing with other families facing the same journey you are on while also doing everything in your power to give your loved one a chance. Even here, right at the beginning, you feel the stress of this process. But you also feel invigorated because, in a situation where it seems like you can do nothing to make it better, you are able to do SOMETHING. You made a promise to provide the best care possible and this is how you are going to do it. You double down on your resolve and keep emailing.
The more likely process for rare disease families is to stalk the clinical trial website daily investigating the listed trials, trying desperately to understand unfamiliar research terminology and gain an unofficial medical degree overnight. You watch for “Active, not recruiting” to change to “Recruiting.” You call and email the clinical trial coordinators and then call and email again when they don’t respond back to you soon enough. Then you have your local doctors call and email on your behalf, hoping they will get a response. You struggle to understand, when your loved one is literally dying a little more every second of every day, how clinical trial administrators can just react without your sense of life-or-death urgency. You curse the very guidelines and bureaucracy that you know are designed to keep participants safe for delaying the one thing that could actually make a difference in your loved one’s prognosis. You see the limited number of spots available and feel guilty about competing with other families facing the same journey you are on while also doing everything in your power to give your loved one a chance. Even here, right at the beginning, you feel the stress of this process. But you also feel invigorated because, in a situation where it seems like you can do nothing to make it better, you are able to do SOMETHING. You made a promise to provide the best care possible and this is how you are going to do it. You double down on your resolve and keep emailing.
And let’s say you make it—you get the call or the email telling you that your loved one is eligible for pre-screening as we did. You are excited but what comes next isn’t easy either. It means putting your child through more bloodwork and having doctors do and send more paperwork until finally you get the go ahead to travel to the trial site for the official screening process. When we were invited to screen, we needed to find childcare for our daughter, take time off of work, and travel across country with a child with special needs. We then had to put our son through more medical procedures along with day-long cognitive and developmental testing. We did this all with the constant reminder that only 11 participants were being admitted into this trial. Would our son be one of them?
The Choice
When going to screen for a clinical trial, there are actually two evaluations happening simultaneously. The first and obvious evaluation is that the clinical trial team is assessing the potential participant to see if the individual meets the criteria to be included in the trial. Second, the participants (and their family) are evaluating the trial to see if it meets their expectations. Or at least they should be.
The power differential is very obvious in rare disease clinical trial evaluation. Because the disease is rare, families don’t have multiple options from which to choose. And because these rare diseases can often be fatal or life-limiting, families don’t have the time to wait for more options to become available. Often, due to the cost of experimental treatments for rare disease, participation slots in the trial are limited. This raises the urgency rare disease families feel when being offered a chance for a clinical trial and puts the majority of the power in the hands of the clinical trial provider. When families feel this heightened urgency and sense of competition for inclusion in the trial, it is difficult for them to have the necessary space and resources available to assess their risk tolerances. To families, the trial treatment could very well be the difference between life and death for their loved one. It’s a great deal of pressure to put on a family already living the nightmare of a difficult diagnosis.
Receiving an offer of a clinical trial screening opened up a door to a level of hope that we hadn’t even allowed ourselves to contemplate. Upon arriving in Minnesota from Texas, we were overwhelmed by the situation. We were still freshly diagnosed and reeling from the thought that our active 4-year old was actually slowing dying from a terminal genetic disease that we had unwittingly passed on to him. Will had always been a healthy kid overall, so we were unprepared for the toll the multiple medical tests, developmental tests, and hours in hospital rooms would take on us during the evaluation. There were so many questions to consider, so many terms to understand. However, even as we faced these emotional challenges, we would also feel remorse about feeling overwhelmed. It was hard to navigate our feelings regarding the clinical trial evaluation because it felt like questioning a huge gift. We were being SCREENED FOR A CLINICAL TRIAL barely 2 months after diagnosis. We felt the full weight of that privilege. We knew how many other families would give anything to be where we were right then, being evaluated for a CHANCE at a potentially different outcome for our child. How could we turn down such an opportunity?
 There are assumptions about rare disease families within the clinical trial space. It is presumed we will jump at the chance of participating in a clinic trial and immediately embrace being participants if offered the opportunity. Many of these assumptions come from a well-meaning belief that these families will participate in clinical trials because it is the only hope their loved ones have of an improved quality of life. However, what that improved quality of life looks like is up to the families, not to the researchers. There are so many things to consider, not the least of which is that a clinical trial is just that: a trial, and one that fulfills multiple definitions of that word. First, it is a trial in that it is an experiment. Neither the participants nor the researchers can say for certainty that the trial will help; no one knows the outcome. No one knows if it will help, or if it will do nothing, or if it will hurt, or if it will cause issues that might be worse than the issues already expected with the course of the disease. Secondly, it is a trial because it is a test of faith and endurance through subjecting oneself through hardship. This includes the participant and the support network of the participant. Trials require medical care and testing, potential side effects, travel, expense, personal documentation…the list goes on. Participating in a trial is a job within itself; it is an endeavor and one that can burn people out quickly if their expectations from the trial are not met.
There are assumptions about rare disease families within the clinical trial space. It is presumed we will jump at the chance of participating in a clinic trial and immediately embrace being participants if offered the opportunity. Many of these assumptions come from a well-meaning belief that these families will participate in clinical trials because it is the only hope their loved ones have of an improved quality of life. However, what that improved quality of life looks like is up to the families, not to the researchers. There are so many things to consider, not the least of which is that a clinical trial is just that: a trial, and one that fulfills multiple definitions of that word. First, it is a trial in that it is an experiment. Neither the participants nor the researchers can say for certainty that the trial will help; no one knows the outcome. No one knows if it will help, or if it will do nothing, or if it will hurt, or if it will cause issues that might be worse than the issues already expected with the course of the disease. Secondly, it is a trial because it is a test of faith and endurance through subjecting oneself through hardship. This includes the participant and the support network of the participant. Trials require medical care and testing, potential side effects, travel, expense, personal documentation…the list goes on. Participating in a trial is a job within itself; it is an endeavor and one that can burn people out quickly if their expectations from the trial are not met.
When assumptions and expectations do not align, it can lead to a discord between the family and the clinical trial providers, especially if a family initially chooses to be part of a clinical trial without having the time, space, and guidance to fully evaluate their decision. Rare disease families can become disillusioned and decide the burden of the trial is too much to bear. They may experience regret and ultimately decide to leave the trial, which then puts the study itself in jeopardy. It is a delicate balance. It is important we afford rare disease families the space to assess their dignity of risk; that is, allowing them their right to determine what level of risk they are willing to take on that fulfills their definition of quality of life. This could be that they decide to live with the risk of a clinical trial or that they decide to live with the risk of their condition, untreated. Whatever decision a family makes, it must be met with support.
In our case, we sat with our options upon returning home to Texas from Minnesota as we waited to hear if Will would be officially invited to participate in his trial. We reviewed literature, talked with experts, and just held our son, the happy little boy who was completely oblivious that there was anything different about him. When the call came, telling us that Will was a fit for the trial and asking us if we would like to go forward, we said yes.
The Challenge
 When we said “yes” to our son, Will, participating in a clinical trial for Sanfilippo Syndrome, we thought we understood what we had chosen. We had read through the informed consent, asked questions, consulted with medical professionals, and reached out to other families. In our situation, we decided that the known risk of the untreated condition was greater than the potential risk associated with being part of the clinical trial. We also felt a responsibility to the Sanfilippo community. Through Will’s participation, we would be helping push the research forward, possibly to a feasible treatment, that would help all the other children who couldn’t be in this trial.
When we said “yes” to our son, Will, participating in a clinical trial for Sanfilippo Syndrome, we thought we understood what we had chosen. We had read through the informed consent, asked questions, consulted with medical professionals, and reached out to other families. In our situation, we decided that the known risk of the untreated condition was greater than the potential risk associated with being part of the clinical trial. We also felt a responsibility to the Sanfilippo community. Through Will’s participation, we would be helping push the research forward, possibly to a feasible treatment, that would help all the other children who couldn’t be in this trial.
However, the reality of being a part of a clinical trial is that you can never understand what you are choosing until you have lived it. The full clinical trial experience is not something for which you can be fully prepared, especially when it’s a clinical trial for a rare condition. There are unique practical and emotional challenges involved. There are sacrifices.
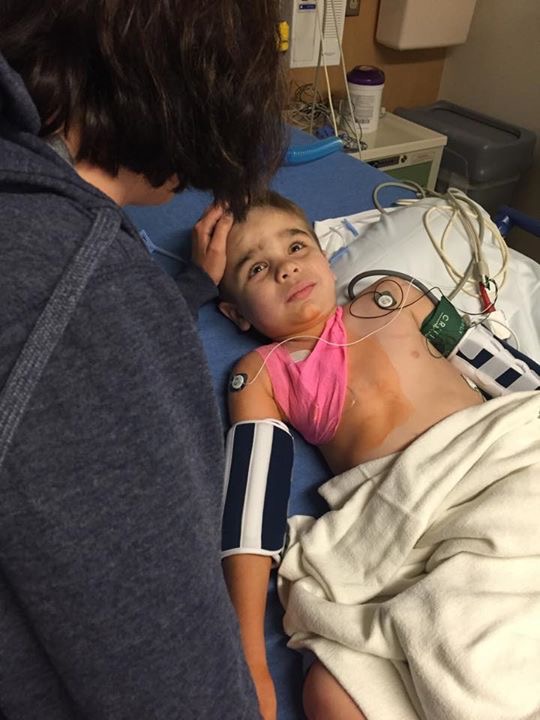 Trials for rare conditions often only occur in select locations due to the limited patient population and the cost that goes into procuring trial sites. This places the burden of either routine traveling or even relocation on a family, taking them away from their usual support networks. For us, Will’s trial required biweekly trips to Minnesota from Texas. Every other Wednesday, Will and I would pack our bags, head to the airport, eat lunch, fly north, pick up a rental car, pick up dinner, and check into our hotel. The next morning, we would eat breakfast, pack up, check-out, and head to the hospital for a full day. At roughly dinner time, we’d be discharged, head to the airport, return the rental car, find dinner at the airport, and fly back home in time to get Will to bed for school on Friday. It was exhausting. Caring for a child with special needs is taxing in normal conditions, but to do it when traveling and by oneself is overwhelming.
Trials for rare conditions often only occur in select locations due to the limited patient population and the cost that goes into procuring trial sites. This places the burden of either routine traveling or even relocation on a family, taking them away from their usual support networks. For us, Will’s trial required biweekly trips to Minnesota from Texas. Every other Wednesday, Will and I would pack our bags, head to the airport, eat lunch, fly north, pick up a rental car, pick up dinner, and check into our hotel. The next morning, we would eat breakfast, pack up, check-out, and head to the hospital for a full day. At roughly dinner time, we’d be discharged, head to the airport, return the rental car, find dinner at the airport, and fly back home in time to get Will to bed for school on Friday. It was exhausting. Caring for a child with special needs is taxing in normal conditions, but to do it when traveling and by oneself is overwhelming.
Because of the effects of Sanfilippo on executive processes, Will never had a sense of “danger” and had no impulse control. For his safety, I could never leave him alone, not even for me to go to the bathroom or to take a shower. I remember intentionally dehydrating myself until we got to the hospital, where I could have a nurse stay with him for brief periods so I could use the restroom. But there was no other option. My husband could not take off from work the amount of time necessary to accompany me and my friends and family had jobs and children of their own of which to take care. For our quarterly week-long trips, I was able to secure help, but for our 2-day adventures, Will and I were on our own.
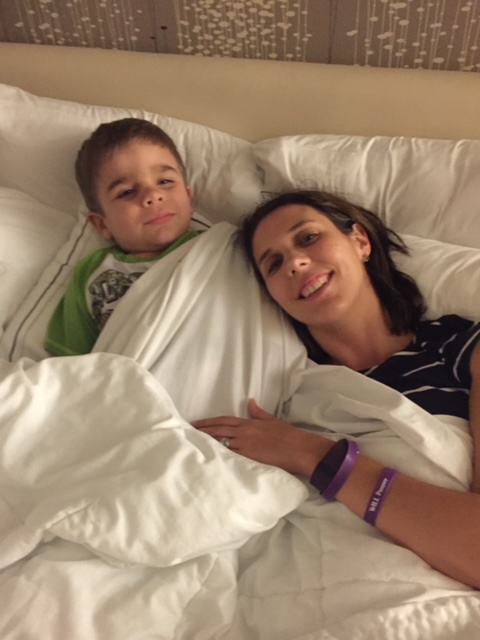 But being on our own was not necessarily a bad thing. It was a special time for he and I to be together. I cherish many of our memories as we worked to make Minnesota more like a home-away-from-home than just a trial location. We made friends with employees at the car rental desk, at our hotel, and at the restaurants we frequented. Airport staff knew Will by name, recognizing him quickly due to our purple WILLPower sweatshirts and his signature traveling hat, a fedora. Hospital staff knew exactly when to turn Paw Patrol or Daniel Tiger on the TV to prevent Will from becoming fussy during procedures. We met other families in our trial and supported each other, as we were the only ones who could really understand what we were all facing. These little things helped lighten the load.
But being on our own was not necessarily a bad thing. It was a special time for he and I to be together. I cherish many of our memories as we worked to make Minnesota more like a home-away-from-home than just a trial location. We made friends with employees at the car rental desk, at our hotel, and at the restaurants we frequented. Airport staff knew Will by name, recognizing him quickly due to our purple WILLPower sweatshirts and his signature traveling hat, a fedora. Hospital staff knew exactly when to turn Paw Patrol or Daniel Tiger on the TV to prevent Will from becoming fussy during procedures. We met other families in our trial and supported each other, as we were the only ones who could really understand what we were all facing. These little things helped lighten the load.
However, these bright spots didn’t erase the other challenges we faced. Being part of the clinical trial meant that even our home life was a data set. Any scrape, bump, doctor visit, or medication had to be reported. Also, due to the clinical trial time commitment, I was unable to work, meaning our family budget needed adjusted to account for being based on a single income for the foreseeable future. It also meant that we would have to continue to procure regular childcare for our toddler daughter so that I could travel with Will and so my husband could continue to work to provide for our family.
These practical challenges led to emotional costs. We were already dealing with the emotional costs of the disease itself, but the trial also added anxiety as we worried whether the treatment was helping Will or not. Traveling added stress to a marriage that was already feeling the weight of having a child with a terminal diagnosis. Leaving my daughter every other week was very difficult and emotional for me as a mother and losing my career-focus was very difficult and emotional for me as a professional. Our family would have benefited from counseling, but time and money were now stretched even tighter due to trial participation.
We worked together, we rallied, and we did all that was required of us. We were faithful to our agreement to the trial and felt that as long as we continued to put in the work, we were giving Will his best shot at a high quality of life.
And then our trial was cancelled.
The Cancellation
I remember the day so well. Will was 2 years into the 3-year enzyme replacement trial for Sanfilippo Syndrome and I was at the airport, for once not bound for Minnesota. Instead, I was alone and heading to visit a dear friend in Illinois. It was to be a much needed and much anticipated weekend of respite for me. It felt like a huge splurge, this gift of a weekend away from caregiving duties. However, a caregiver can’t ever truly set aside the job of caregiving. Walking through the airport by myself was disorienting and I found myself having small moments of panic when I forgot that I wasn’t traveling with Will, frantically glancing around for him before remembering that I was alone. My thoughts were so used to being caught up in his care that I couldn’t comprehend the privilege of drinking coffee while it was still hot or being able to go to the bathroom by myself.
As I told myself to focus, to take in this gift and allow myself to rest, my phone rang.
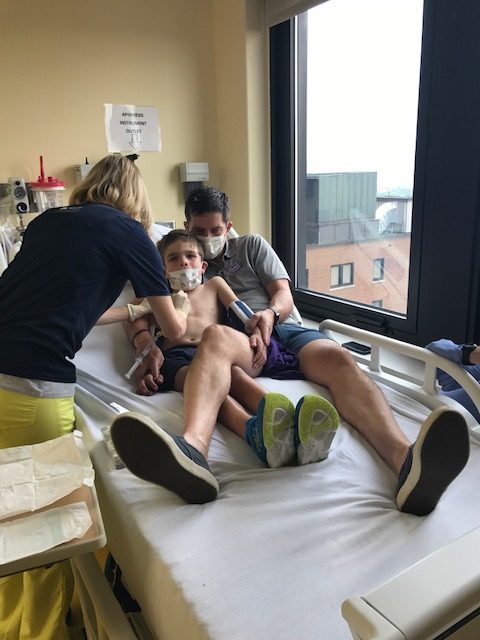 Although we had read a press release earlier in the year telling us that the trial sponsor wasn’t looking to expand Will’s trial due to financial constraints, the news delivered in that call was completely unexpected. We had seen the promising data that had been presented at conferences and we had also seen the promising data in our own son. His regression had slowed and his quality of life was high. An expensive treatment, it didn’t come as a surprise that they weren’t adding in more participants, but we had been assured Will would complete the 3-year treatment period. We had even been given the schedule for the start of our third year and had begun work on making all the arrangements that were necessitated by that. So, I was completely caught off guard when I answered the call from another parent from the trial telling me that the trial had been cancelled because the sponsoring company said it wasn’t meeting its goal of stopping the cognitive regression.
Although we had read a press release earlier in the year telling us that the trial sponsor wasn’t looking to expand Will’s trial due to financial constraints, the news delivered in that call was completely unexpected. We had seen the promising data that had been presented at conferences and we had also seen the promising data in our own son. His regression had slowed and his quality of life was high. An expensive treatment, it didn’t come as a surprise that they weren’t adding in more participants, but we had been assured Will would complete the 3-year treatment period. We had even been given the schedule for the start of our third year and had begun work on making all the arrangements that were necessitated by that. So, I was completely caught off guard when I answered the call from another parent from the trial telling me that the trial had been cancelled because the sponsoring company said it wasn’t meeting its goal of stopping the cognitive regression.
I recall feeling as if I was underwater. Shapes moved more slowly around me and sounds were muffled. I think I accidentally knocked into a woman as I walked and, as she yelled at me, I gazed at her without comprehension of what she was saying. I called our clinical trial nurse for confirmation that the trial was actually being ended and, when I had it, I called my husband with a voice filled with panicked tears. “What are we going to do?” I asked, “They are taking it away from him. He’s going to die.”
I was near hysterics, uncomprehending of what was happening and feeling like I was in a dreamlike sequence. I felt just as bad, if not worse, than our diagnosis day. Diagnosis day robbed us of our innocence, but the trial cancellation threatened to rob us of our hope. We knew that losing this treatment was the end of the road for Will. Now considered “damaged goods” by the research community due to having already been part of an experimental treatment, we were told he would never be eligible for a clinical trial again. The possibility of the trial being cancelled prior to the 3-year mark when there hadn’t been any safety issues hadn’t even crossed our minds. We felt abandoned.
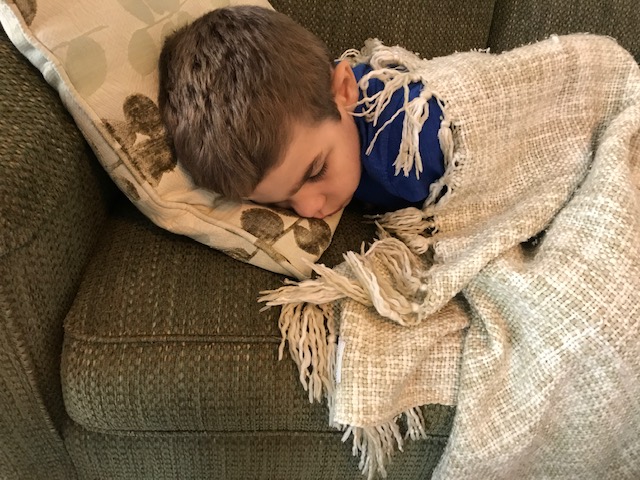 We bore it as well as we could. We had rare disease advocates fight for continued access to the remaining drug and were denied by the pharmaceutical company. We watched our son go through hellacious withdrawal symptoms after being made to quit the infusion cold-turkey as they refused even our request to wean the participating children slowly off of the treatment. Will stopped walking and talking and started screaming in agitation and pain. The clinical trial team, which had required us to report any bump or bruise Will experienced over the past two years, wouldn’t even take our calls or help us navigate the withdrawal period.
We bore it as well as we could. We had rare disease advocates fight for continued access to the remaining drug and were denied by the pharmaceutical company. We watched our son go through hellacious withdrawal symptoms after being made to quit the infusion cold-turkey as they refused even our request to wean the participating children slowly off of the treatment. Will stopped walking and talking and started screaming in agitation and pain. The clinical trial team, which had required us to report any bump or bruise Will experienced over the past two years, wouldn’t even take our calls or help us navigate the withdrawal period.
It took a month but, thankfully, Will started to rebound. His body began to remember how to work on its own again without the regular enzyme infusions it had come to rely on for 2 years. But even then, we continued to feel betrayed by our clinical trial experience. For 2 years, we had allowed our son to be poked, prodded, anesthetized, and examined. He’d endured blood draws and PICC line placements, MRIs and spinal taps. As parents, we’d filled out form after form, assessment after assessment. We’d taken time away from our families and from our work. Then, not only was the clinical trial treatment suddenly taken away, but the trial sponsor refused to share any of the valuable data they had gathered. That shattered us even further. We had chosen to be part of the trial, not only for Will’s benefit, but for the benefit of the Sanfilippo patient community. We knew the data would be kept private during the trial, but we had incorrectly assumed the data would be published at the end of the trial, regardless of the results. Any data collected from the trial had the potential to be beneficial to other research studies but since the company owned data, they didn’t have to share it. It was like a slap in the face.
 Cancelled trials and the lack of data sharing can have a strong impact on rare disease research: They erode the faith the rare disease community has in the clinical trial process. Families, willing to sacrifice anything to be part of the trial, do not feel valued for their sacrifice. When it feels as though the research community does not have our loved ones’ best interests at heart, why should we subject ourselves or our children to the substantial risks and burdens of clinical trials? And yet without clinical trials, how do we fulfill our promise to do whatever necessary to give our loved ones the quality of the life they deserve? It can make families feel like they are between a rock and a hard place and alone in our decision making.
Cancelled trials and the lack of data sharing can have a strong impact on rare disease research: They erode the faith the rare disease community has in the clinical trial process. Families, willing to sacrifice anything to be part of the trial, do not feel valued for their sacrifice. When it feels as though the research community does not have our loved ones’ best interests at heart, why should we subject ourselves or our children to the substantial risks and burdens of clinical trials? And yet without clinical trials, how do we fulfill our promise to do whatever necessary to give our loved ones the quality of the life they deserve? It can make families feel like they are between a rock and a hard place and alone in our decision making.
The Change
Following the cancellation of Will’s trial, we continued our efforts to raise the profile of Sanfilippo Syndrome. However, due to our recent clinical trial experience, we had new advocacy avenues we wanted to explore and support, changes we wanted to see made. Recognizing that there was a disconnect between the clinical trial endpoints prioritized by pharmaceutical companies and the clinical trial endpoints prioritized by rare disease patients and their caregivers, we worked with the Cure Sanfilippo Foundation as they developed their Caregiver Preference Study to present to the FDA. This study, based on responses from actual caregivers, highlighted the need to include clinical trial endpoints that were based around quality of life improvements that were important to families. The goal is now for any proposed clinical trial for Sanfilippo Syndrome to include these factors as significant endpoints to help determine the success level of a potential treatment. We further worked with the Foundation to support other clinical trial changes, including open data sharing, increased aftercare following a trial’s completion, and more caregiver input and involvement throughout the clinical trial process.
 The Cure Sanfilippo Foundation prioritized these clinical trial recommendations when they developed and funded their own quality of life clinical trial, the Open-label Study of Anakinra in MPS III. Although not designated as a potential “cure” for Sanfilippo Syndrome, this study was designed with goal of mitigating some of the symptoms of Sanfilippo in order to improve quality of life for both the patients and the caregivers. The design of the study also took caregiver burden into account and wrote in specific protocols to make participating in the trial as family-friendly as possible. This trial is an important model demonstrating how research and family priorities do not have to be in conflict with one another and how, by including patient and caregiver voices in clinical trial development, trial sponsosr can devise treatment options that better meet the needs of patients while also building bridges for continued trust and cooperation within rare disease communities.
The Cure Sanfilippo Foundation prioritized these clinical trial recommendations when they developed and funded their own quality of life clinical trial, the Open-label Study of Anakinra in MPS III. Although not designated as a potential “cure” for Sanfilippo Syndrome, this study was designed with goal of mitigating some of the symptoms of Sanfilippo in order to improve quality of life for both the patients and the caregivers. The design of the study also took caregiver burden into account and wrote in specific protocols to make participating in the trial as family-friendly as possible. This trial is an important model demonstrating how research and family priorities do not have to be in conflict with one another and how, by including patient and caregiver voices in clinical trial development, trial sponsosr can devise treatment options that better meet the needs of patients while also building bridges for continued trust and cooperation within rare disease communities.
The most important aspect of this new trial for us? It didn’t exclude children who had been in clinical trials in the past from applying to participate. And so, again, we had another tough decision to make. Knowing what we now knew about the clinical trial process, would we attempt to do it again? Our questions this time were much more focused, making sure to assume nothing and get every detail necessary to make an informed decision. We now DID know what we were getting ourselves into and wanted to make sure all the concerns we’d had in the past were being addressed. Because this trial had been developed with caregiver preferences and support in mind, we felt much more comfortable with applying for screening.
 And so, in 2020, Will again donned his purple WILLPower shirt and traveling hat to this time head to California, where he was screened for another clinical trial. He was accepted and began his second clinical trial for Sanfilippo Syndrome in summer of 2020. This is another previously inconceivable phenomenon in the rare disease world: the opportunity for a second trial. But hopefully, as we continue to elevate the importance of quality of life benefits in rare disease research and educate companies on how to best work with rare disease patients and their families, this concept will become less unusual, giving more people the chance fulfill their promise to do anything they can to give their loved one more good days.
And so, in 2020, Will again donned his purple WILLPower shirt and traveling hat to this time head to California, where he was screened for another clinical trial. He was accepted and began his second clinical trial for Sanfilippo Syndrome in summer of 2020. This is another previously inconceivable phenomenon in the rare disease world: the opportunity for a second trial. But hopefully, as we continue to elevate the importance of quality of life benefits in rare disease research and educate companies on how to best work with rare disease patients and their families, this concept will become less unusual, giving more people the chance fulfill their promise to do anything they can to give their loved one more good days.
________________________________________________________________________________________________________________
 Valerie Tharp Byers, EdD is a psychology and education researcher who lives in Spring, Texas with her husband, Tim, and children, Will and Samantha. Valerie became a rare disease advocate in 2015 following the diagnosis of Will with Sanfilippo Syndrome, a degenerative and fatal genetic disorder. Valerie is a Board Member of the Cure Sanfilippo Foundation, where she works to raise both the public profile of Sanfilippo Syndrome and the funds necessary to support research and clinical trials. Valerie and Tim also co-founded and operate WILLPower, an organization in their local area dedicated to raising Sanfilippo Syndrome awareness.
Valerie Tharp Byers, EdD is a psychology and education researcher who lives in Spring, Texas with her husband, Tim, and children, Will and Samantha. Valerie became a rare disease advocate in 2015 following the diagnosis of Will with Sanfilippo Syndrome, a degenerative and fatal genetic disorder. Valerie is a Board Member of the Cure Sanfilippo Foundation, where she works to raise both the public profile of Sanfilippo Syndrome and the funds necessary to support research and clinical trials. Valerie and Tim also co-founded and operate WILLPower, an organization in their local area dedicated to raising Sanfilippo Syndrome awareness.
